What is in this leaflet
This leaflet answers some common questions about BOSENTAN VIATRIS.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What BOSENTAN VIATRIS is used for
BOSENTAN VIATRIS is used for the treatment of high blood pressure in the blood vessels between the heart and the lungs. This condition is called pulmonary arterial hypertension.
This medicine acts to reduce abnormally high blood pressure by widening these blood vessels. It belongs to the class of medicines known as endothelin receptor antagonists.
Your doctor however, may prescribe BOSENTAN VIATRIS for another purpose.
Ask your doctor if you have any questions about why it has been prescribed for you.
This medicine is only available with a doctor’s prescription.
Before you take BOSENTAN VIATRIS
When you must not take it
Do not take this medicine if you are:
-
Pregnant or intend to become pregnant. You must stop taking the medicine at least 3 months before trying to become pregnant.
It is known that this medicine causes harm to the developing baby if you take it during pregnancy and in the three months before becoming pregnant. -
Breastfeeding
Tell your doctor immediately if you are breastfeeding. You are advised to stop breastfeeding if this medicine is prescribed for you because it is not known if this drug passes into the milk in women who are taking this medicine. - being treated with cyclosporine A (a medicine used after a transplant or to treat psoriasis)
- being treated with glibenclamide (a medicine used for diabetes)
Do not take BOSENTAN VIATRIS if you are allergic to it or any of the ingredients listed at the end of this leaflet.
Do not take BOSENTAN VIATRIS if you have moderate or severe liver disorder.
Do not take it after the expiry date (EXP) printed on the pack. If you take it after the expiry date has passed, it may not work as well.
Do not take it if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take BOSENTAN VIATRIS
BOSENTAN VIATRIS may harm sperm. All men should use effective birth control while taking this medicine and for 3 months after they stop taking it.
If sexually active, you must use a hormonal and barrier method of contraception. This medicine may reduce the effectiveness of hormone contraceptives such as the pill and hormone patches, implants or injection. It is important to use other contraceptives, like condoms or an intrauterine device.
Your doctor will advise you about using reliable contraceptives before taking this medicine.
You must have a negative pregnancy test at the time of starting treatment if you are sexually active. Your doctor will need evidence that you are not pregnant.
Tell your doctor if:
- You are a woman of childbearing potential and not using reliable contraceptive methods. You must have a negative pregnancy test before beginning treatment. The test should be performed on the second day of a normal menstrual period or 11 days after the last unprotected sexual intercourse, whichever is later. Your doctor will advise you about using reliable contraception before taking or whilst taking this medicine.
Hormonal contraception on its own is not a reliable option because this medicine may make this method ineffective in preventing pregnancy. Hormonal contraceptives include ones you take orally (the pill), patches you put on your skin, ones that are injected and implants. You should ALWAYS use additional methods of contraception such as condoms and IUDs and not rely just on hormonal contraception. You should have a pregnancy test every month while you are taking this medicine. You must stop taking this medicine for at least 3 months prior to becoming pregnant.
-
you are breastfeeding or planning to breastfeed.
It is not known whether BOSENTAN VIATRIS passes into breast milk. - You have allergies to:
- any other medicines
- any other substances such as foods, preservatives or dyes.
- You have or have had any medical conditions, especially the following:
- pulmonary arterial hypertension
- anaemia
- hypotension
- liver or renal disorders
- heart failure
- HIV infection
If you have not told your doctor about any of the above, tell them before you use BOSENTAN VIATRIS.
While taking BOSENTAN VIATRIS
Do not become pregnant while taking this medicine.
You must have a pregnancy test every month while you are taking this medicine. Your doctor will need evidence that you are not pregnant before prescribing this medicine again.
Taking other medicines
Tell your doctor if you are taking any other medicines including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with BOSENTAN VIATRIS. These include:
- hormonal contraceptives (oral, injectable, transdermal and implantable)
- simvastatin a medicine for lowering blood fats
- medicines for diabetes such as glibenclamide
- medicines for fungal infections such as ketoconazole, fluconazole and voriconazole
- medicines for bacterial infections such as rifampicin
- medicine to prevent organ transplantation rejection such as cyclosporine A, tacrolimus and sirolimus
- medicines for rheumatoid arthritis or psoriasis/dermatitis
- lopinavir+ritonavir or other ritonavir-boosted protease inhibitors (used to treat HIV)
- digoxin (used to treat heart rhythm disorders)
- sildenafil or tadalafil (used to treat erectile dysfunction and/or pulmonary hypertension)
- anticonvulsant medicines (used to treat seizures) such as carbamazepine, phenobarbital (phenobarbitone) and phenytoin
- St John's wort, a herbal preparation sometimes used to treat depression
These medicines may be affected by BOSENTAN VIATRIS or may affect how well it works. You may need to use different amounts of your medicines, or take different medicines.
Your doctor will advise you.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking BOSENTAN VIATRIS.
How to take BOSENTAN VIATRIS
How much to take
Always take this medicine exactly as your doctor has instructed you. You should check with your doctor or pharmacist if you are unsure.
The usual dose is one tablet, twice daily. For the first 4 weeks you will take a 62.5 mg tablet twice daily. Depending on how you respond to the medicine, your doctor may increase the dosage after four weeks to a 125 mg tablet twice daily.
Talk to your doctor if you do not think the medicine is working or you think it is working too well. Your doctor may need to change the dose you are taking.
How to take it
BOSENTAN VIATRIS is taken by mouth, in the morning and evening, and can be taken with or without food.
How long to take it
Do not stop taking this medicine unless your doctor tells you to. Stopping your treatment may lead to a worsening of your symptoms. Your doctor may tell you to reduce the dose over a few days before stopping completely.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take the next dose when you are meant to.
Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting an unwanted side effect.
If there is still a long time to go before your next dose, take it as soon as you remember and then go back to taking it as you would normally.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (Australia telephone 13 11 26; New Zealand telephone 0800 POISON or 0800 764 766) for advice, or go to Accident and Emergency at your nearest hospital if you think that you or anyone else may have taken too much BOSENTAN VIATRIS.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using BOSENTAN VIATRIS
Things you must do
It is very important that you have a liver function blood test before you start treatment and every month after that. BOSENTAN VIATRIS can cause liver damage if it is not found early. Because this side effect may not cause symptoms at first, only a blood test can show that you have early liver damage. Regular blood tests let your doctor change or stop your therapy before there is permanent damage.
You should have a blood test for anaemia after 1 and 3 months, and then every 3 months for the rest of your treatment.
You need to have regular pregnancy tests if you are a female of childbearing age.
Things you must not do
Do not give the tablets to anyone else even if they have the same symptoms as you.
Things to be careful of
If you feel dizzy whilst taking this medicine, do not drive or operate machinery.
Side effects
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- nausea
- shortness of breath or difficulty breathing
- vomiting
- fever
- unusual tiredness
- stomach pain
- yellowing of your skin or the whites of your eyes (jaundice)
These may be serious side effects of BOSENTAN VIATRIS. You may need urgent medical attention.
Serious side effects are uncommon.
Other side effects of this medicine include:
- headache
- inflamed throat and irritated nose passages
- flushing (hot flashes)
- ankle and leg swelling
- low blood pressure
- blood disorders
- fast heart beat
- tiredness
- itching, rash, skin inflammation, skin redness
- diarrhoea
See your doctor if any of these worry you.
Tell your doctor if you notice anything else that is making you feel unwell. Other side effects not listed above may occur in some consumers.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After taking BOSENTAN VIATRIS
Storage
Keep your tablets in the bottle until it is time to take them. If you take the tablets out of the bottle they may not keep well.
Store this medicine in the original bottle, in a cool, dry place where the temperature stays below 25°C.
Do not store BOSENTAN VIATRIS or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
BOSENTAN VIATRIS 62.5 mg tablets are white to off-white, film-coated, round, biconvex, beveled edge tablet debossed with "M" on one side of the tablet and "BN1" on the other side.
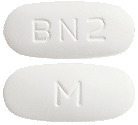
BOSENTAN VIATRIS 125 mg tablets are white to off-white, film-coated, oval, biconvex, beveled edge tablet debossed with "M" on one side of the tablet and "BN2" on the other side.
Both strengths are available in HDPE bottles of 60 tablets.
Ingredients
Active ingredient:
BOSENTAN VIATRIS 62.5 mg: 62.5 mg bosentan (as monohydrate) per tablet.
BOSENTAN VIATRIS 125 mg: 125 mg bosentan (as monohydrate) per tablet.
Inactive ingredients:
- Maize starch
- Pregelatinised maize starch
- Sodium starch glycollate
- Povidone
- Glyceryl behenate
- Magnesium stearate
- Sodium lauryl sulfate
- OPADRY complete film coating system 03K580000 WHITE (ARTG PI No: 108721)
- SURELEASE aqueous ethylcellulose dispersion E-7-19040 clear (ARTG PI No. 11522)
BOSENTAN VIATRIS also contains traces of sulfites.
Supplier
BOSENTAN VIATRIS is supplied in Australia by:
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
Phone: (02) 9298 3999
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in June 2022.
Australian registration numbers:
BOSENTAN VIATRIS 125 mg:
AUST R 206776
BOSENTAN VIATRIS_cmi\Feb23/00
Published by MIMS April 2023