Prodeinextra®
| Consumer Medicine Information (CMI) summary |
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
| 1. Why am I using Prodeinextra? |
Prodeinextra contains the active ingredients paracetamol and codeine phosphate hemihydrate. Prodeinextra is used to relieve acute moderate pain and fever. For more information, see Section 1. Why am I using Prodeinextra? in the full CMI.
| 2. What should I know before I take Prodeinextra? |
Do not take if you have ever had an allergic reaction to Prodeinextra or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Prodeinextra? in the full CMI.
| 3. What if I am taking other medicines? |
Some medicines may interfere with Prodeinextra and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
| 4. How do I take Prodeinextra? |
The standard dose for adults and children aged 12 years and over for this medicine is 2 caplets, taken every 4 to 6 hours if necessary.
More instructions can be found in Section 4. How do I use Prodeinextra? in the full CMI.
| 5. What should I know while using Prodeinextra? |
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines | Be careful before you drive or use any machines or tools until you know how Prodeinextra affects you. Prodeinextra may cause dizziness, drowsiness or light-headedness in some people. |
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Prodeinextra? in the full CMI.
| 6. Are there any side effects? |
- Tell your doctor or pharmacist immediately if you notice any of the following side effects shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue, or other parts of the body, rash, itching or hives on the skin. They may be the signs of an allergic reaction.
- Tell your doctor or pharmacist if you notice any of the following and they worry you: nausea or vomiting, constipation, drowsiness or dizziness. These are the more common side effects of your medicine. They are usually mild.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
| WARNING: Limitations of use Prodeinextra should only be used if your doctor decides other treatment options are not able to effectively manage your pain or you cannot tolerate them. Hazardous and harmful use Prodeinextra contains codeine which may be habit forming. Prodeinextra poses risks of abuse, misuse and addiction which can lead to overdose and death. Your doctor will assess your risks and monitor you regularly during treatment. Life threatening respiratory depression Serious, life-threatening, or fatal respiratory depression (shallow or difficulty breathing) may occur with the use of Prodeinextra even when used as recommended. These problems can occur at any time during use, but the risk is higher when first starting Prodeinextra and after a dose increase, if you are older, or have an existing problem with your lungs. Your doctor will monitor you and change the dose as appropriate. Use of other medicines while using Prodeinextra Using Prodeinextra with other medicines that can make you feel drowsy such as sleeping tablets (e.g. benzodiazepines), other pain relievers, antihistamines, antidepressants, antipsychotics, gabapentinoids (e.g. gabapentin and pregabalin), cannabis and alcohol may result in severe drowsiness, decreased awareness, breathing problems, coma and death. Your doctor will minimize the dose and duration of use and monitor you regularly for signs and symptoms of breathing difficulties and sedation. You must not drink alcohol while taking Prodeinextra. |
Prodeinextra®
Active ingredient(s): paracetamol and codeine phosphate hemihydrate
| Consumer Medicine Information (CMI) |
This leaflet provides important information about using Prodeinextra. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Prodeinextra.
Where to find information in this leaflet:
1. Why am I using Prodeinextra?
2. What should I know before I take Prodeinextra?
3. What if I am taking other medicines?
4. How do I take Prodeinextra?
5. What should I know while using Prodeinextra?
6. Are there any side effects?
7. Product details
| 1. Why am I using Prodeinextra? |
Prodeinextra contains the active ingredients paracetamol and codeine phosphate hemihydrate.
Prodeinextra is a type of analgesic. Paracetamol and codeine work together to stop the pain messages from getting through to the brain. Paracetamol also acts in the brain to help reduce fever.
Prodeinextra is used to relieve acute moderate pain and fever and is intended for short term use.
| 2. What should I know before I take Prodeinextra? |
Warnings
Do not take Prodeinextra if:
- you are allergic to paracetamol or codeine phosphate hemihydrate, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have severe and/or acute respiratory diseases
- you have unstable asthma or emphysema, respiratory depression (shallow breathing) or respiratory insufficiency (difficulty breathing).
- you have bronchitis
- you have liver failure
- you have Glucose-6-phosphate-dehydrogenase deficiency (an enzyme deficiency)
- you are a known CYP 2D6 ultra-rapid metaboliser (a fast metaboliser of codeine by the CYP 2D6 enzyme)
- you have a history of intolerance to this medicine
- you have diarrhoea caused by antibiotics or poisoning
Do not take Prodeinextra if you have a history of drug dependence, including alcohol dependence.
Do not take Prodeinextra during the third trimester of pregnancy.
Do not take it if you are in labour, especially if the baby is premature.
Do not take Prodeinextra if you are allergic to morphine or oxycodone.
Do not give Prodeinextra to children under 12 years.
Do not give Prodeinextra to children aged between 12-18 years who have undergone tonsillectomy and/or adenoidectomy to treat sleep apnoea.
Do not take it if you are breastfeeding or planning to breastfeed.
Do not take it after the expiry date (EXP) printed on the pack. If you take it after the expiry date has passed, it may not work as well.
Do not take it if the packaging is damaged or shows signs of tampering.
Check with your doctor or pharmacist if you have or have had any of the following medical conditions:
- heart, lung, liver or kidney problems
- low blood pressure
- difficulty breathing, wheezing, chronic cough, asthma, or other chronic breathing conditions
- compromised respiratory function (due to emphysema, kyphoscoliosis or obesity)
- known analgesic intolerance
- a history of drug dependence, including alcohol dependence
- pre-existing opioid dependence
- chronic alcohol use including recent cessation of alcohol intake
- low glutathione reserves
- Gilbert's syndrome
- stomach, gallbladder, urinary or bowel conditions
- chronic constipation
- recent stomach, intestines or urinary tract surgery
- prostate problems
- problems with your adrenal gland or thyroid
- multiple sclerosis
- head injury or trauma
- convulsions, fits or seizures.
Tell your doctor or pharmacist if you plan to have surgery.
Tell your doctor or pharmacist if you drink large quantities of alcohol.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Do not take Prodeinextra during the third trimester of pregnancy.
Do not take it if you are in labour, especially if the baby is premature.
Prodeinextra contains codeine, which may produce withdrawal effects in the newborn baby.
Do not take it if you are breastfeeding or planning to breastfeed.
Prodeinextra passes into breast milk and there is a possibility your baby may be affected.
Addiction
You can become addicted to Prodeinextra even if you take it exactly as prescribed. Prodeinextra may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Dependence
As with all other opioid containing products, your body may become used to you taking Prodeinextra. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Prodeinextra suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance
Tolerance to Prodeinextra may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
Prodeinextra given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
| 3. What if I am taking other medicines? |
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may affect how Prodeinextra works. These include:
- medicines causing sleepiness or drowsiness
- tranquillisers (medicines for anxiety and nerves)
- gabapentinoids, hypnotics, cannabis and centrally-active anti-emetics
- benzodiazepines (medicines used as sedatives or to treat anxiety)
- medicines containing alcohol (ethanol), e.g. some cough syrups
- medicines used to treat depression
- anticholinergics (medicines for stomach cramps/spasms, travel sickness, Parkinson's disease)
- antipsychotics (medicines used to treat mental illnesses)
- monoamine oxidase inhibitors (medicines used to treat depression) taken within the last 10 days
- salicylates or non-steroidal antiinflammatory drugs (NSAIDS), such as aspirin or ibuprofen
- medicines which thin the blood
- medicines to treat epilepsy
- other pain relief medication
- medicines used to treat high blood pressure
- medicines used to relax muscles
- medicines used to treat diarrhoea, nausea or vomiting
- propantheline, a medicine used to treat stomach ulcers
- cholestyramine (medicine used to treat bile problems and/or high cholesterol)
- chelating resin
- medicines used to treat alcohol and/or opioid dependence (e.g. naltrexone, buprenorphine or methadone)
- chloramphenicol (medicine used to treat ear and eye infections)
- flucloxacillin, zidovudine or rifampicin (medicines used to treat infections).
- medicines used to control electrolytes levels in kidney disease
These medicines may be affected by Prodeinextra, or may affect how well it works. You may need to use different amounts of your medicine, or take different medicines. Your doctor or pharmacist will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Prodeinextra.
| 4. How do I take Prodeinextra? |
How much to take
- The standard dose for adults and children aged
12 years and over for this medicine is 2 caplets, taken every 4 to 6 hours if necessary. - Follow the instructions provided.
- Do not exceed the recommended dosage.
- You should not take more than 8 caplets in a 24 hour period.
Ask your doctor or pharmacist if you are unsure of the correct dose for you.
They will tell you exactly how much to take.
If you take the wrong dose, Prodeinextra may not work as well and your problem may not improve.
How to take Prodeinextra
- Swallow the caplets whole with a little water or other liquid.
When to take Prodeinextra
- If you are not sure when to take it, ask your doctor or pharmacist.
How long to take Prodeinextra
Do not take this medicine for longer than 48 hours (for children aged 12-17 years) or for more than 3 days, except on medical advice.
- Ask your doctor or pharmacist if you are not sure how long to take the medicine for.
If you forget to take Prodeinextra
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you take too much Prodeinextra
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately call triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used Prodeinextra that was prescribed for you. If someone takes an overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heartbeat
- Nausea or vomiting
- Convulsions or fits
If you think that you have taken too much Prodeinextra, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
If you take too much Prodeinextra, you will probably feel nauseous, experience stomach pain, sweating, anxiety, light-headed, dizzy, or drowsy.
Depending on your body's individual ability to break down codeine, you may experience signs of overdose even when you take Prodeinextra as recommended by your doctor. If overdose symptoms occur, seek immediate medical advice.
| 5. What should I know while using Prodeinextra? |
Things you should do
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are taking this medicine.
If you become pregnant while you are taking given this medicine, stop taking it and tell your doctor or pharmacist immediately.
Remind any doctor, dentist or pharmacist you visit that you are using Prodeinextra.
Things you should not do
- Do not take more than the recommended dose unless your doctor tells you to.
Adults and children over 12 years should not take more than 8 caplets a day.
Do not take high doses of the medicine for long periods of time unless your doctor tells you to. Taking more than the recommended dose may cause liver damage. Codeine may be habit forming.
- Do not give this medicine to anyone else, even if they have the same condition as you.
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Prodeinextra affects you.
Prodeinextra may cause dizziness, drowsiness or light-headedness and problems with vision in some people, especially after the first dose.
Children should not ride bikes if affected and should be supervised to avoid potential harm.
Drinking alcohol
Do not drink alcohol while taking Prodeinextra.
The effects of alcohol could be made worse while taking Prodeinextra. It is not recommended that you drink alcohol while taking Prodeinextra.
Looking after your medicine
- Store below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it. A locked cupboard at least one-and-a half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
| 6. Are there any side effects? |
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Gastrointestinal-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Metabolism related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
| 7. Product details |
This medicine is only available with a doctor's prescription.
What Prodeinextra contains
| Active ingredient (main ingredient) | Paracetamol and codeine phosphate hemihydrate. |
| Other ingredients (inactive ingredients) | Maize starch, purified talc, pregelatinised maize starch, povidone, stearic acid, potassium sorbate, magnesium stearate and microcrystalline cellulose. |
| Potential allergens | Contains potassium sorbate. Prodeinextra does not contain gluten, sucrose, lactose, tartrazine or any other azo dyes. |
Do not take this medicine if you are allergic to any of these ingredients.
What Prodeinextra looks like
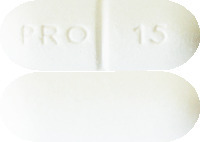
Prodeinextra are white to off-white capsule-shaped tablets (caplets) marked “PRO 15” and scored on one side with a plain reverse (AUST R 205550).
Prodeinextra is available in packs of 24 and 40* caplets.
*pack size not marketed.
Who distributes Prodeinextra
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
Toll Free Number (medical information): 1800 818 806
Email: medinfo.australia@sanofi.com
This leaflet was prepared in December 2021
prodeine-ccdsv5-cmiv13-03dec21
Published by MIMS January 2022