Consumer medicine information
Triumeq Tablets
Dolutegravir + Abacavir + Lamivudine
BRAND INFORMATION
Brand name
Triumeq
Active ingredient
Dolutegravir + Abacavir + Lamivudine
Schedule
S4
Triumeq Tablets
Boxed Warnings
Abacavir, a component of Triumeq tablets, is associated with hypersensitivity reactions, which can be life-threatening, and in rare cases fatal. Triumeq tablets, or any other medicinal product containing abacavir (Kivexa [abacavir/lamivudine], Trizivir [abacavir/lamivudine/zidovudine] and Ziagen [abacavir]) must never be restarted following a hypersensitivity reaction (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
1 Name of Medicine
Dolutegravir (as sodium), abacavir (as sulfate) and lamivudine.
2 Qualitative and Quantitative Composition
Triumeq film-coated tablets contain 50 mg of dolutegravir (as dolutegravir sodium), 600 mg of abacavir as abacavir sulfate and 300 mg of lamivudine. Product Information for Tivicay (dolutegravir), Ziagen (abacavir), 3TC (lamivudine) and Kivexa (abacavir and lamivudine) contain additional information.
Dolutegravir sodium is a white to light yellow powder and is slightly soluble in water.
Abacavir sulfate is a white to off-white crystalline powder with a solubility of approximately 77 mg/mL in water at 25°C.
Lamivudine is a white to off-white crystalline solid which is highly soluble in water.
Triumeq tablets also contain mannitol. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
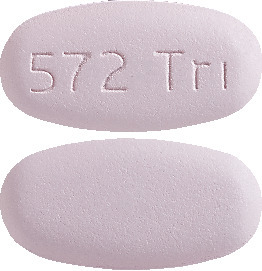
Purple, film-coated, oval, biconvex tablets, debossed with '572 Tri' on one side. Each film-coated tablet contains 50 mg of dolutegravir (as dolutegravir sodium), 600 mg of abacavir (as abacavir sulphate) and 300 mg of lamivudine.
4 Clinical Particulars
4.9 Overdose
Limited experience of single higher doses (up to 250 mg in healthy patients) revealed no specific symptoms or signs, apart from those listed as adverse reactions.
No specific symptoms or signs have been identified following acute overdose with abacavir or lamivudine, apart from those listed as adverse reactions.
If overdose occurs, the patient should be treated supportively with appropriate monitoring as necessary. Since lamivudine is dialysable, continuous haemodialysis could be used in the treatment of overdose, although this has not been studied. It is not known whether abacavir can be removed by peritoneal dialysis or haemodialysis. As dolutegravir is highly bound to plasma proteins, it is unlikely that it will be significantly removed by dialysis.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.3 Preclinical Safety Data
Dolutegravir was not mutagenic or clastogenic using in vitro tests in bacteria and cultured mammalian cells, and an in vivo rodent micronucleus assay.
Neither abacavir nor lamivudine were mutagenic in bacterial tests, but both induced mutations in a mouse lymphoma assay and were clastogenic in human peripheral lymphocytes in vitro. In rats, lamivudine did not cause chromosomal damage in bone marrow cells in vivo or cause DNA damage in primary hepatocytes. Abacavir was clastogenic in an in vivo micronucleus assay in mice, but not in rats when tested in combination with lamivudine at systemic exposures corresponding to 86 and 31 times the clinical exposure level for ABC and 3TC, respectively.
Dolutegravir was not carcinogenic in long term studies in the mouse and rat (respectively, 27 and 23 times the 50 mg human clinical exposure when dolutegravir is administered in combination with abacavir and lamivudine, based on AUC).
Carcinogenicity studies with orally administered abacavir in mice and rats showed an increase in the incidence of malignant and non-malignant tumours. Malignant tumours occurred in the preputial gland of males and the clitoral gland of females of both species, and in the liver, urinary bladder, lymph nodes and the subcutis of female rats. Nonmalignant tumours occurred in the liver of mice and rats, Harderian gland of female mice, and thyroid gland of rats. In rats, there were also increased incidences of urothelial hyperplasia and urinary bladder tumours, associated with increased urinary calculi.
The majority of these tumours occurred at the highest abacavir dose of 330 mg/kg/day in mice and 600 mg/kg/day in rats. These dose levels were equivalent to 21 to 30 times the expected systemic exposure in humans when abacavir is administered in combination with dolutegravir and lamivudine. The exception was preputial gland tumours in mice which occurred at a dose of 110 mg/kg. Exposure at this dose is approximately 5 times the expected human systemic exposure. The carcinogenic potential in humans is unknown.
When lamivudine was administered orally to separate groups of rodents at doses up to 2000 times (mice and male rats) and 3000 (female rats) mg/kg/day, there was no evidence of a carcinogenic effect due to lamivudine in the mouse study. In the rat study there was an increased incidence of endometrial tumours at the highest dose (approximately 70 times the estimated human exposure at the recommended therapeutic dose of one tablet twice daily, based on AUC). However, the relationship of this increase to treatment is uncertain.
Mild myocardial degeneration in the heart of mice and rats was observed following administration of abacavir for two years. The systemic exposures were equivalent to 7 to 19 times human exposure at 600 mg when abacavir is administered in combination with dolutegravir and lamivudine. The clinical relevance of this finding has not been determined.
6 Pharmaceutical Particulars
6.7 Physicochemical Properties
The chemical (IUPAC) name for dolutegravir sodium is Sodium (4R,12aS)-9-{[(2,4-difluorophenyl)methyl]carbamoyl}- 4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H- pyrido[1',2':4,5]pyrazino [2,1-b][1,3]oxazin-7-olate. It has a molecular formula of C20H18F2N3NaO5 and a molecular weight of 441.36 g/mol. The partition coefficient (log P) for dolutegravir sodium is 2.2 and the pKa is 8.2.
The chemical name of abacavir sulfate is (1S,cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol sulfate (salt) (2:1). Abacavir sulfate is the enantiomer with 1S,4R absolute configuration on the cyclopentene ring. It has a molecular formula of (C14H18N6O)2.H2SO4 and a molecular weight of 670.76 g/mol.
The chemical name of lamivudine is (2R,cis)-4-amino-1-[2- (hydroxymethyl)-1,3-oxathiolan-5-yl]-2(1H)-pyrimidinone. Lamivudine is the (-)enantiomer of a dideoxy analogue of cytidine. Lamivudine has also been referred to as (-)2',3'-dideoxy, 3'-thiacytidine. It has a molecular formula of C8H11N3O3S and a molecular weight of 229.3 g/mol.
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSDOLUTE.gif
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSABASUL.gif
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSLAMIVU.gif
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes
https://stagingapi.mims.com/au/public/v2/images/fulltablegif/TRIUMEST.gif
