1 Name of Medicine
Vosevi (sofosbuvir/velpatasvir/voxilaprevir 400 mg/100 mg/100 mg) tablets.
2 Qualitative and Quantitative Composition
Vosevi is available as a fixed-dose combination tablet. The active substances in Vosevi tablets are sofosbuvir, velpatasvir and voxilaprevir.
Each tablet contains 400 mg of sofosbuvir, 100 mg of velpatasvir, and 100 mg of voxilaprevir.
Sofosbuvir is a white to off-white powder with a solubility of ≥ 2 mg/mL across the pH range of 2-7.7 at 37°C.
Velpatasvir is a white to tan or yellow solid. Velpatasvir is practically insoluble (less than 0.1 mg/mL) above pH 5, slightly soluble (3.6 mg/mL) at pH 2, and soluble (greater than 36 mg/mL) at pH 1.2.
Voxilaprevir is a white to light brown solid. It is slightly hygroscopic to hygroscopic. Voxilaprevir is practically insoluble (less than 0.1 mg/mL) below pH 6.8.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
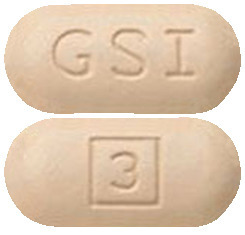
Vosevi tablets are beige, capsule-shaped, film coated tablets, debossed with "GSI" on one side and "3" on the other side.
4 Clinical Particulars
4.9 Overdose
The highest documented doses of sofosbuvir, velpatasvir, and voxilaprevir were single doses of 1200 mg, 500 mg, and 900 mg, respectively. In healthy volunteer studies with sofosbuvir and velpatasvir, there were no untoward effects observed at these dose levels, and adverse events were similar in frequency and severity to those reported in the placebo groups. The most common adverse events in patients receiving voxilaprevir 900 mg were diarrhoea (34%), vomiting (19%), and nausea (17%). The effects of higher doses/exposures are not known.
No specific antidote is available for overdose with Vosevi. If overdose occurs the patient must be monitored for evidence of toxicity. Treatment of overdose with Vosevi consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient. Hemodialysis can efficiently remove the predominant circulating metabolite of sofosbuvir, GS-331007, with an extraction ratio of 53%. Hemodialysis is unlikely to result in significant removal of velpatasvir or voxilaprevir since velpatasvir and voxilaprevir are highly bound to plasma proteins.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia) and 0800 764 766 (New Zealand).
5 Pharmacological Properties
5.3 Preclinical Safety Data
Genotoxicity. Sofosbuvir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes, and in vivo mouse micronucleus assays.
Velapatasvir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes, and in vivo rat micronucleus assays.
Voxilaprevir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes and in vivo rat micronucleus assays.
Carcinogenicity. Carcinogenicity studies in mice and rats do not indicate any carcinogenicity potential of sofosbuvir administered at doses up to 200 mg/kg/day in male mice and 600 mg/kg/day in female mice, and 750 mg/kg/day in rats. Exposure to GS-331007 in these studies in mice was up to 4 times (male) and 17 times (female), and in rats up to 8 times (male) and 10 times (female) higher than the clinical exposure at 400 mg sofosbuvir.
Velpatasvir was not carcinogenic in a 26-week transgenic mouse study at exposures up to 42 and 67 times higher than human exposure in male and female mice, respectively. A carcinogenicity study in rats is ongoing.
Carcinogenicity studies for voxilaprevir have not been conducted.
6 Pharmaceutical Particulars
6.7 Physicochemical Properties
Chemical structure. Sofosbuvir is a nucleotide inhibitor of HCV NS5B RNA-dependent RNA polymerase, velpatasvir is a Hepatitis C Virus (HCV) NS5A inhibitor and voxilaprevir is an HCV NS3/4A protease inhibitor.
The chemical name of sofosbuvir is (S)-Isopropyl 2-((S)-(((2R,3R,4R,5R)- 5-(2,4-dioxo-3,4-dihydropyrimidin-1 (2H)-yl)-4-fluoro-3-hydroxy-4 -methyltetrahydrofuran-2-yl) methoxy)-(phenoxy)phosphorylamino) propanoate. It has a molecular formula of C22H29FN3O9P and a molecular weight of 529.45. It has the following structural formula:
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSSOFOSB.gif The chemical name of velpatasvir is Methyl {(1R)-2-[(2S,4S)- 2-(5-{2-[(2S,5S)- 1-{(2S)-2-[(methoxycarbonyl) amino]-3-methylbutanoyl} -5-methylpyrrolidin-2-yl] -1,11-dihydro[2] benzopyrano[4',3':6,7] naphtho[1,2-d]imidazol -9-yl}-1H-imidazol-2-yl)-4- (methoxymethyl)pyrrolidin- 1-yl]-2-oxo-1-phenylethyl} carbamate. It has a molecular formula of C49H54N8O8 and a molecular weight of 883.0. It has the following structural formula:
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSVELPAT.gif The chemical name for voxilaprevir is (1aR,5S,8S,9S,10R,22aR) -5-tert-butyl-N-{(1R,2R)- 2-(difluoromethyl)-1- [(1-methylcyclopropanesulfonyl) carbamoyl] cyclopropyl}-9-ethyl-18,18-difluoro- 14-methoxy-3,6-dioxo- 1,1a,3,4,5,6,9,10, 18,19,20,21,22,22a-tetradecahydro -8H-7,10-methanocyclopropa [18,19][1,10,3,6] dioxadiazacyclononadecino [11,12-b] quinoxaline-8-carboxamide. It has a molecular formula of C40H52F4N6O9S and a molecular weight of 868.9. It has the following structural formula:
https://stagingapi.mims.com/au/public/v2/images/fullchemgif/CSVOXILA.gif CAS number. Sofosbuvir: 1190307-88-0.
Velpatasvir: 1377049-84-7.
Voxilaprevir: 1535212-07-7.
7 Medicine Schedule (Poisons Standard)
S4.
Summary Table of Changes
https://stagingapi.mims.com/au/public/v2/images/fulltablegif/VOSEVIST.gif